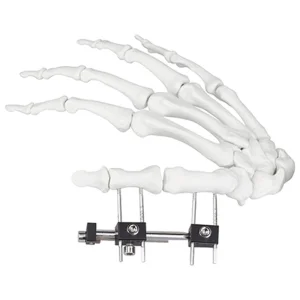
 Orthopedic Surgical Fixation Mini External Fixator Universal Compression Extension Type
Orthopedic Surgical Fixation Mini External Fixator Universal Compression Extension Type
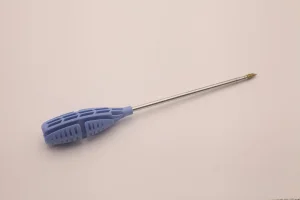 Titanium Suture Anchor for Sport Medicine
Titanium Suture Anchor for Sport Medicine
 Ilizarov External Fixator Foot Ring with CE
Ilizarov External Fixator Foot Ring with CE
 Nx Medical China Factory Titanium Tlif Cage Peek for Lumbar Interbody Fusion
Nx Medical China Factory Titanium Tlif Cage Peek for Lumbar Interbody Fusion
 5.5 Pedicle Screw for Spine Nail-Rod Instrument Sets, Lumbar Posterior Titanium Rod Installation
5.5 Pedicle Screw for Spine Nail-Rod Instrument Sets, Lumbar Posterior Titanium Rod Installation
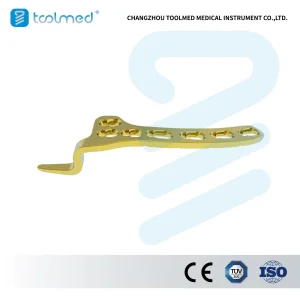 Titanium Surgical Plate Implants Price Clavicle Hook Locking Titanium Plate
Titanium Surgical Plate Implants Price Clavicle Hook Locking Titanium Plate
 High-Strength Cocrmo Thoracolumbar Pedicle Screw and Rod System
High-Strength Cocrmo Thoracolumbar Pedicle Screw and Rod System
 Nickel Plated Floor Heating Radiator Manifold Water Mixing System
Nickel Plated Floor Heating Radiator Manifold Water Mixing System
The global spinal surgery landscape is witnessing a paradigm shift. As a premier OEM/ODM Interbody Fusion Cages Supplier, we recognize that the industry has moved far beyond simple mechanical support. Today, the focus is on Biological Integration and Biomechanical Harmony.
From the early days of stainless steel spacers to the modern ubiquity of PEEK (Polyetheretherketone) and 3D-printed Titanium, the goal remains the same: achieving a successful arthrodesis while minimizing patient morbidity. Modern interbody cages now feature advanced geometries that facilitate bone ingrowth, specialized surface coatings (such as Hydroxyapatite or Plasma-sprayed Titanium), and radiolucent properties that allow for clear post-operative imaging.
Our commitment to Information Gain in the spinal sector involves analyzing clinical outcomes to refine our OEM designs. We don't just manufacture; we innovate by addressing the "stress shielding" effect, ensuring our cages possess a modulus of elasticity closer to human bone, thereby reducing the risk of subsidence and implant failure.
Utilizing Medical Grade 5 Titanium and PEEK-OPTIMA™ for maximum biocompatibility and durability.
Anatomically shaped cages including PLIF, TLIF, ALIF, and LLIF options for diverse surgical approaches.
Porous surface architectures engineered via 3D printing to promote rapid bone fusion.
Wide range of footprints, heights, and lordotic angles to match specific patient anatomy.
International medical device distributors and hospital chains face significant challenges: regulatory hurdles, fluctuating lead times, and the need for consistent quality. Our Interbody Fusion Cage Exporter services are designed to mitigate these risks through a robust supply chain framework.
We provide a "one-stop-shop" for brands looking to expand their spinal portfolio. Our services include initial CAD design, rapid prototyping, mechanical testing (static and dynamic fatigue), and final sterile packaging. By partnering with us, clients gain access to advanced manufacturing technologies without the overhead costs of maintaining their own production lines.
The future of interbody fusion lies in 3D-printed Titanium. Our roadmap focuses on perfecting "trabecular structures" that mimic the porosity of cancellous bone. This allows for immediate mechanical stability and long-term biological fixation.
We are exploring nano-topographies that can influence cellular behavior at the molecular level. By modifying the implant surface, we can accelerate osteoblast differentiation and reduce the risk of biofilm formation.
The integration of sensor technology to monitor fusion progress in real-time is the next frontier. While still in early stages, our R&D department is collaborating with academic institutions to explore the feasibility of "Smart Cages."
Shenzhen Weed Medical Apparatus Co., Ltd., established in 2015, primarily engages in the sales of medical devices and maintains in-depth business and technical cooperation with leading domestic and international consumable manufacturers. Most of its products boast technology and materials superior to industry standards.
Our operation is built on the E-E-A-T principle. With years of experience in the Sichuan Province market—serving over 200 hospitals—we have gathered invaluable clinical data that informs our manufacturing processes. Our technical and sales personnel always pay attention to user needs, opinions, and suggestions during clinical use.









The company collaborates closely with professors, universities, and frontline clinical staff in domestic hospitals. Sales are the company's cash cow. During the sales process, the company consistently focuses on the technological contributions and shortcomings of its products to the industry. Our technical department collaborates with hospital experts, university professors, and manufacturers to develop solutions to common clinical problems.
Adherence to ISO 13485:2016, CE (MDR), and FDA standards. We assist partners in local registrations and documentation.
Strategic warehousing and logistics support to ensure timely delivery across 30+ countries.
Providing surgical technique guides, webinars, and on-site support for orthopedic surgeons.
A: We primarily work with Medical Grade 5 Titanium Alloy (Ti-6Al-4V) and PEEK. We also offer 3D-printed porous titanium and hydroxyapatite-coated options for enhanced fusion.
A: Yes, our ODM service allows for complete customization of lordotic angles (typically 0°, 4°, 8°, 12°, or custom), footprints, and expansion mechanisms to meet specific market requirements.
A: For standard products, shipping usually occurs within 2-4 weeks. For custom OEM projects, the timeline ranges from 8 to 16 weeks depending on design complexity and regulatory needs.
A: While we provide dedicated instrumentation sets, we can design cages to be compatible with existing universal insertion tools used in your target market.
 Pedicle Screw Fixation System Orthopaedic Spinal Implants Posterior Cervical Surgery
Pedicle Screw Fixation System Orthopaedic Spinal Implants Posterior Cervical Surgery
 Unilateral External Fixator-Mini Rail (5mm)
Unilateral External Fixator-Mini Rail (5mm)
 Orthopedic Basic Cage Instruments Set Surgical Instrument
Orthopedic Basic Cage Instruments Set Surgical Instrument
 Orthopedic Distal Tibial Lateral Locking Plates Titanium Orthopedic Implant
Orthopedic Distal Tibial Lateral Locking Plates Titanium Orthopedic Implant
 Advanced Narrow Blade Lamina Hook for Thoracolumbar Fusion
Advanced Narrow Blade Lamina Hook for Thoracolumbar Fusion
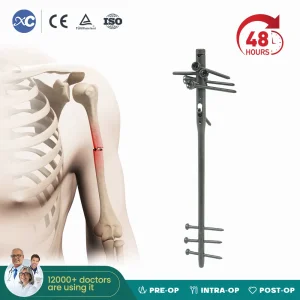 Xc Medico Orthopedic Implant Titanium Interlocking Humerus Nail Multi-Lock
Xc Medico Orthopedic Implant Titanium Interlocking Humerus Nail Multi-Lock
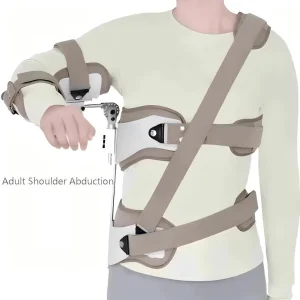 Hot Seller Adult Shoulder Abduction Shoulder Dislocation
Hot Seller Adult Shoulder Abduction Shoulder Dislocation
 Cheap Price Titanium Alloy Coating Surgical Instrument Set Plif Interbody Fusion Peek Cage
Cheap Price Titanium Alloy Coating Surgical Instrument Set Plif Interbody Fusion Peek Cage