

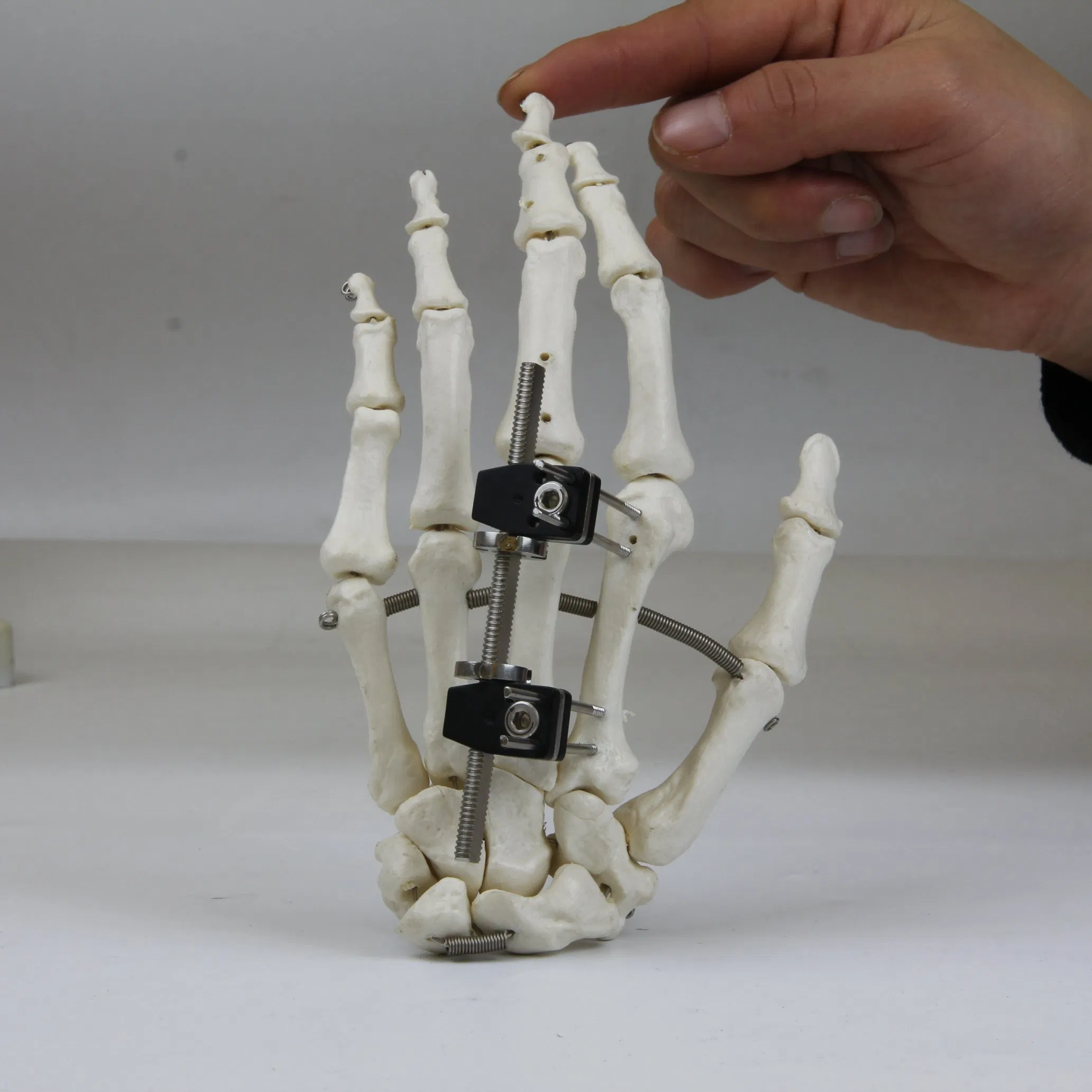












| Customization: | Available |
|---|---|
| Certification: | ISO, CE |
| Condition: | New |




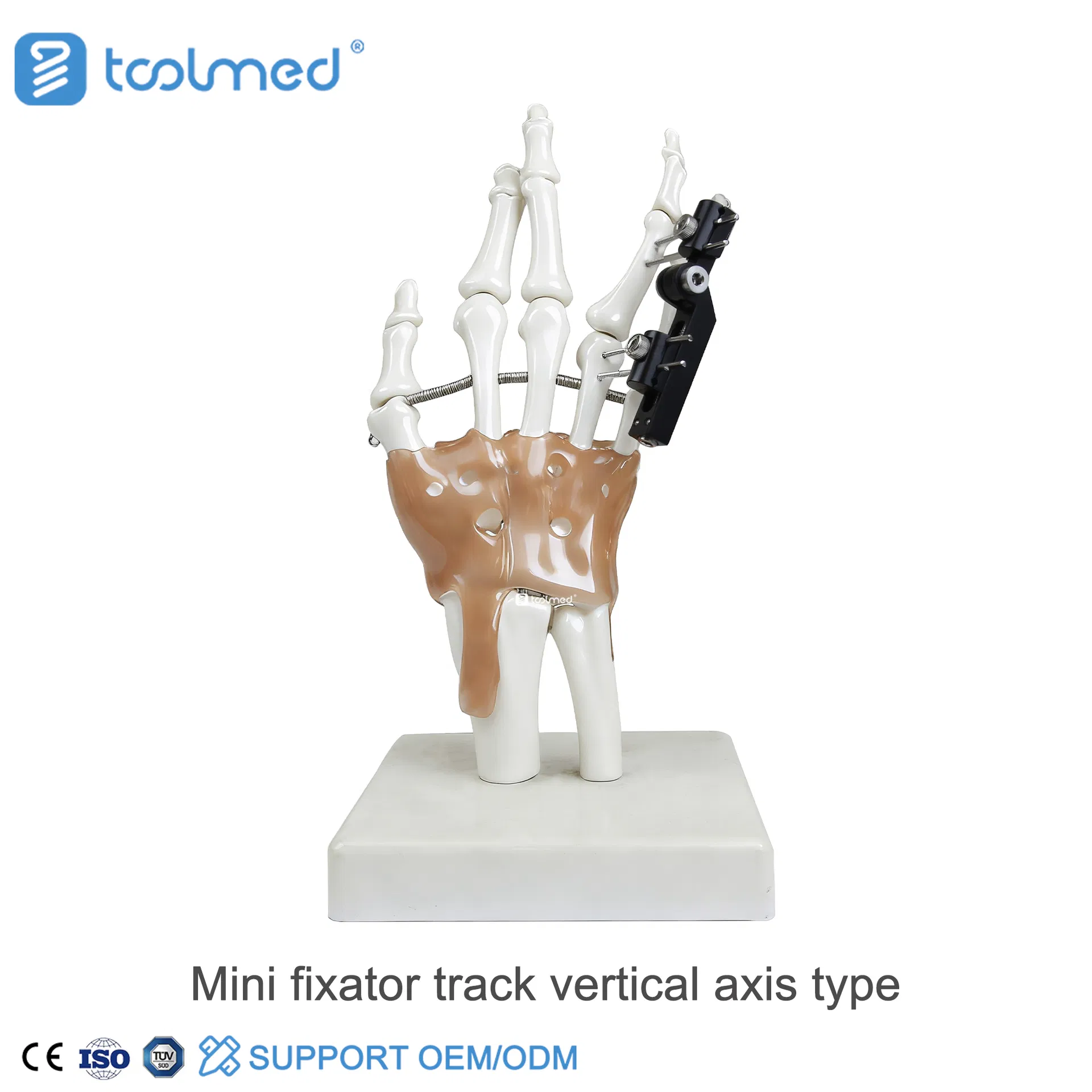
| Model | Product Description | Components | Material |
|---|---|---|---|
| 5200-1206 | Talipes Equinovarus & Equinovalgus & Bone Lengthening External Fixator | Ring 4 PCS, Foot Ring 1 PC, L-connecting 2 PCS, Half-ring 1 PC, Annular Diagonal Tie 1 PC, Inner threaded post 4 PCS, Single joint 2 PCS, M6 threaded rod 8 PCS, Pin clamp 24 PCS | Carbon Fiber/SS |


Founded in 2013, the organization is one of the leading orthopedic manufacturers in China, specializing in the research, design, manufacturing, and sales of orthopedic products including veterinary orthopedic implants and instruments.
The industrial park covers 10,000+ m². With over 100 employees and 60+ CITIZEN & HAAS advanced CNC machines, the facility includes a high-standard laboratory and advanced inspection equipment such as CMM/PP/QV. Operations are conducted under a strict quality system compliant with ISO 9001, ISO 13485, CE, and GMP standards.
We are committed to providing premium and safe products at affordable prices, efficient logistics, dedicated after-sale service, and constant clinical support globally. OEM/ODM services are also available to meet special customer demands. To date, these products have been successfully exported to over 70 countries, receiving excellent recognition and feedback.



 Weed Orthopedic Implants
Weed Orthopedic Implants