











| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |

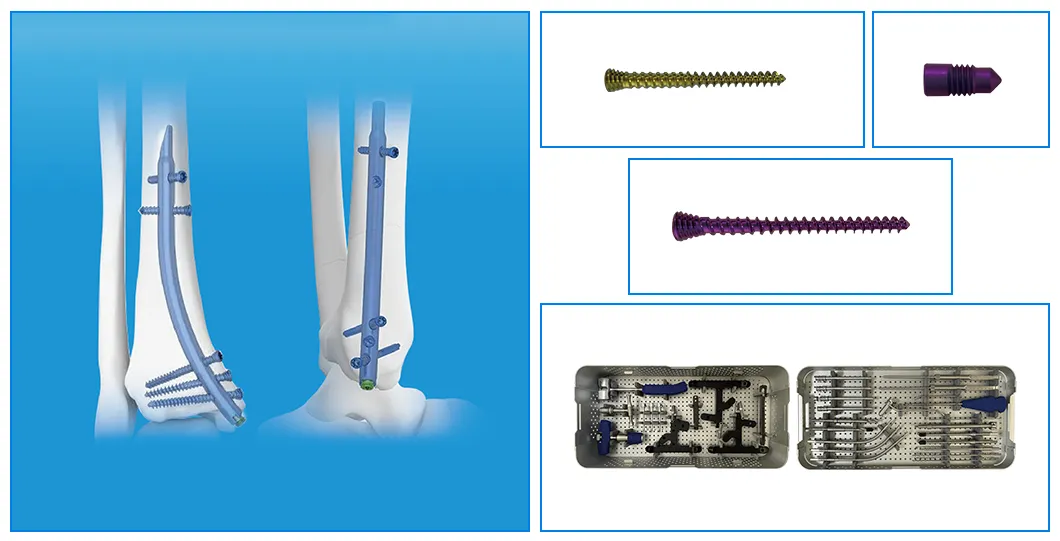
| Product | Size (mm) | Material |
|---|---|---|
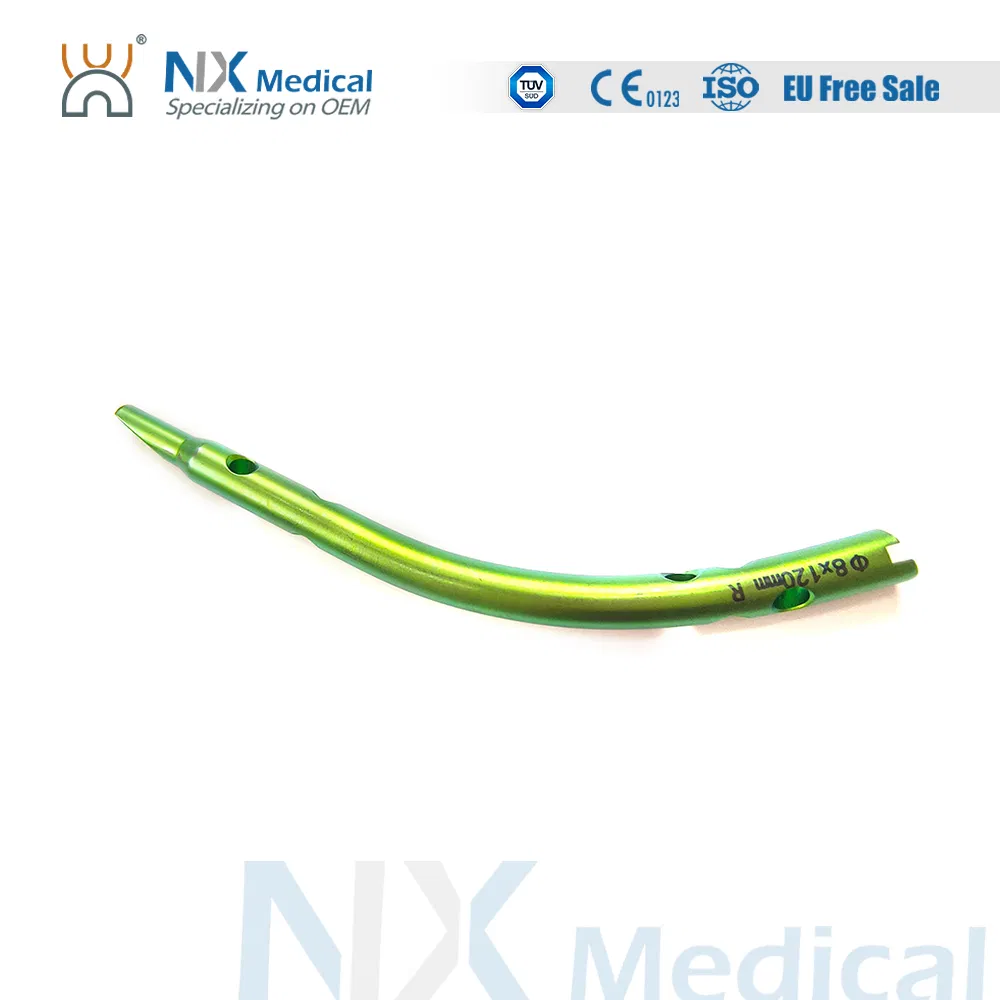
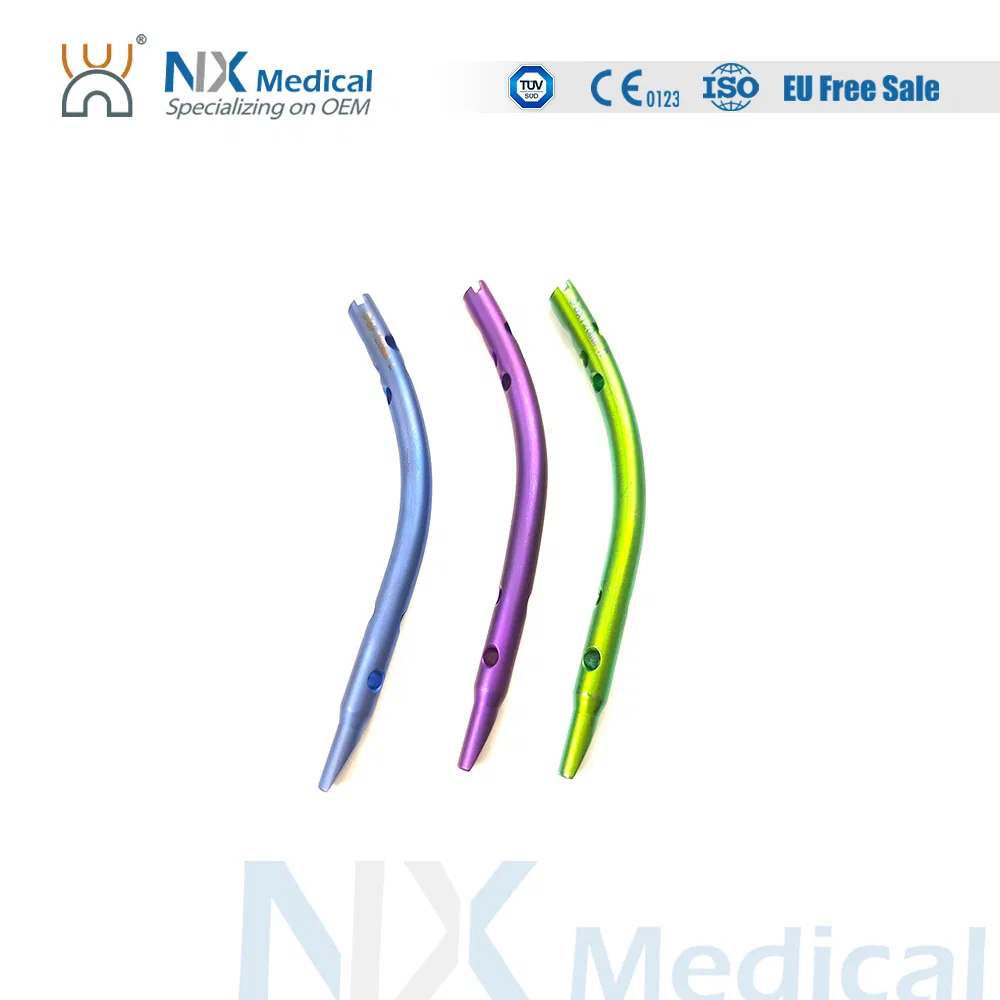
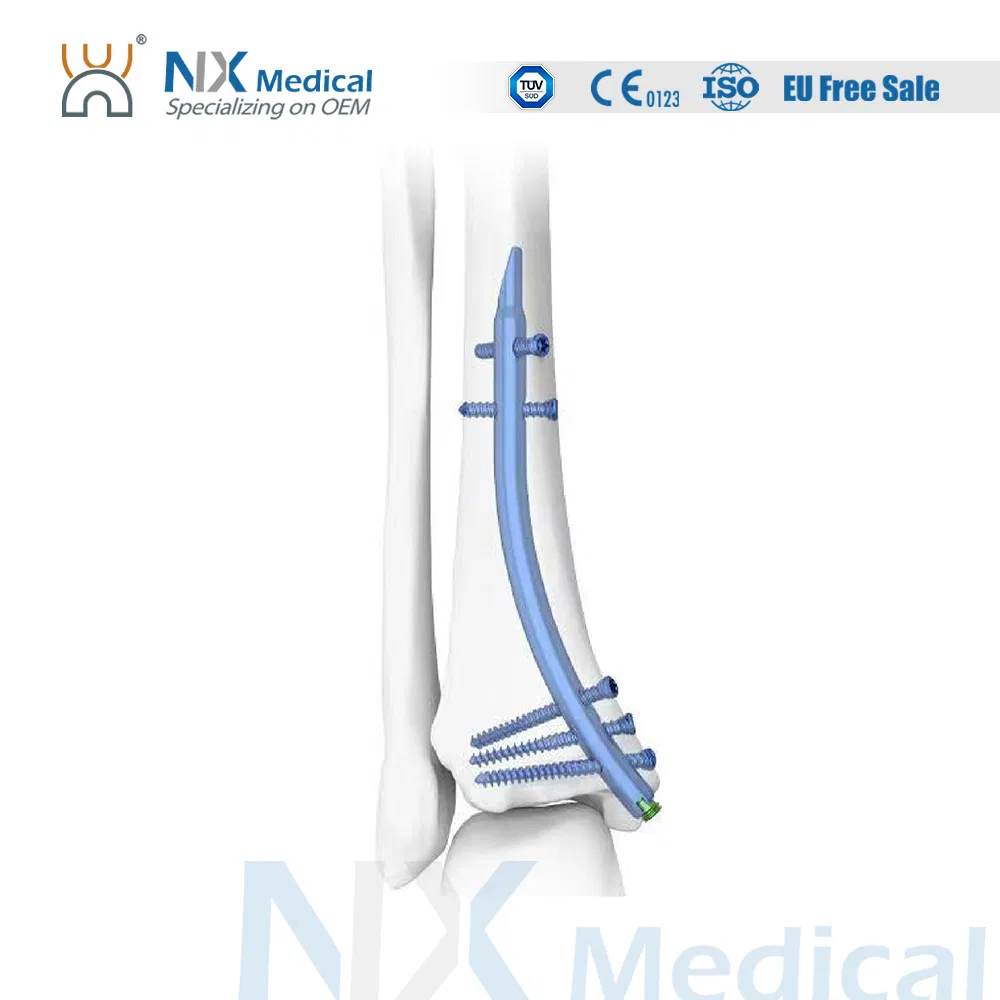
| Retrograde Tibial Nail | 7 x 110 / 8 x 120 / 8 x 140 | Titanium Alloy (TA) |
| φ3.5mm Interlocking Cancellous Screw | 30 / 32 / 34 / 36 / 38 / 40 / 42 / 44 / 46 / 48 / 50 / 52 | Titanium Alloy (TA) |
| φ3.5mm Interlocking Cortical Screw | 18 / 20 / 22 / 24 / 26 / 28 | Titanium Alloy (TA) |
| φ4.0mm Interlocking Cortical Screw | 24 / 26 / 28 / 30 / 32 / 34 | Titanium Alloy (TA) |
| Retrograde Tibial Nail End Cap | Standard / 5 | Titanium Alloy (TA) |
Our company is one of the leading Orthopedic product manufacturers located in the East of China. We provide a comprehensive range of cost-effective surgical and reliable therapeutic solutions to Orthopedic surgeons and patients worldwide. We offer superior and various kinds of Orthopedic trauma implants, spinal implants, and surgical instruments.
For the Orthopedic trauma system, our major categories are locking plates, intramedullary nails, cannulated headless screws, and cable systems. We are committed to the global Orthopedic industry with continuous investment in R&D. We believe superior quality is the key for patients who use our products, and we have established a strict quality system in compliance with ISO13485, TUV CE0123, and NMPA.




 Weed Orthopedic Implants
Weed Orthopedic Implants