




1 / 5










| Still deciding? Get samples of $ ! US$ 113/Piece Request Sample |
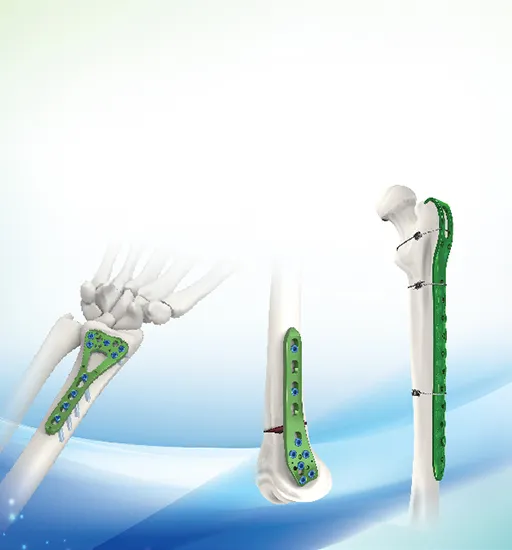
| Proximal Femur Neck with Hook Locking Plate 238 | ||
|---|---|---|
| P/N | Material | Holes |
| 23804-23812 | Titanium | 4,6,8,10,12 holes |
| Use 5.0 locking screw, 5.0 cortical self tapping screw | ||








 Weed Orthopedic Implants
Weed Orthopedic Implants