














| Customization: | Available |
|---|---|
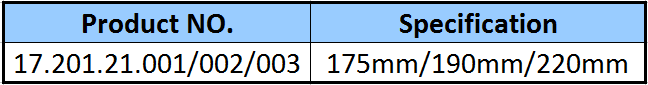
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
Semi-Ring Combined Fixator
Indication: Primarily used for fractures around the hip joint.
Description: Specially designed for tibial plateau fractures and distal tibial fractures. It is employed in conjunction with 3 to 4 olive-head Kirschner wires and a 4.8mm drill bit.
Description of Screws: Available in 6.0/5.0 screws (SS/TA).
As a national high-tech enterprise, R&D investment has consistently exceeded 8% of total sales, enabling the conversion of clinical expertise and innovative ideas into practical achievements. We've launched several groundbreaking products with national invention and utility model patents.
Key revolutionary technologies include: the Rev Drill system, the Three-Row Stapler, Sternal Fixation and Rib Plate.

Core Philosophy: 'Good faith as the base, quality above all.'
The manufacturing process adheres rigorously to the ISO13485 quality system, CE MDD 93/42/EEC directives, and medical device manufacturing quality management specifications.
Equipped with advanced processing centers from Germany and USA, and automatic lathes from Japan. The Testing Center includes a biomechanics laboratory and a physical-chemical testing center for:

Products are exported globally to countries including Colombia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. We also serve as a major OEM supplier for some of the largest global players in the medical industry.









 Weed Orthopedic Implants
Weed Orthopedic Implants