




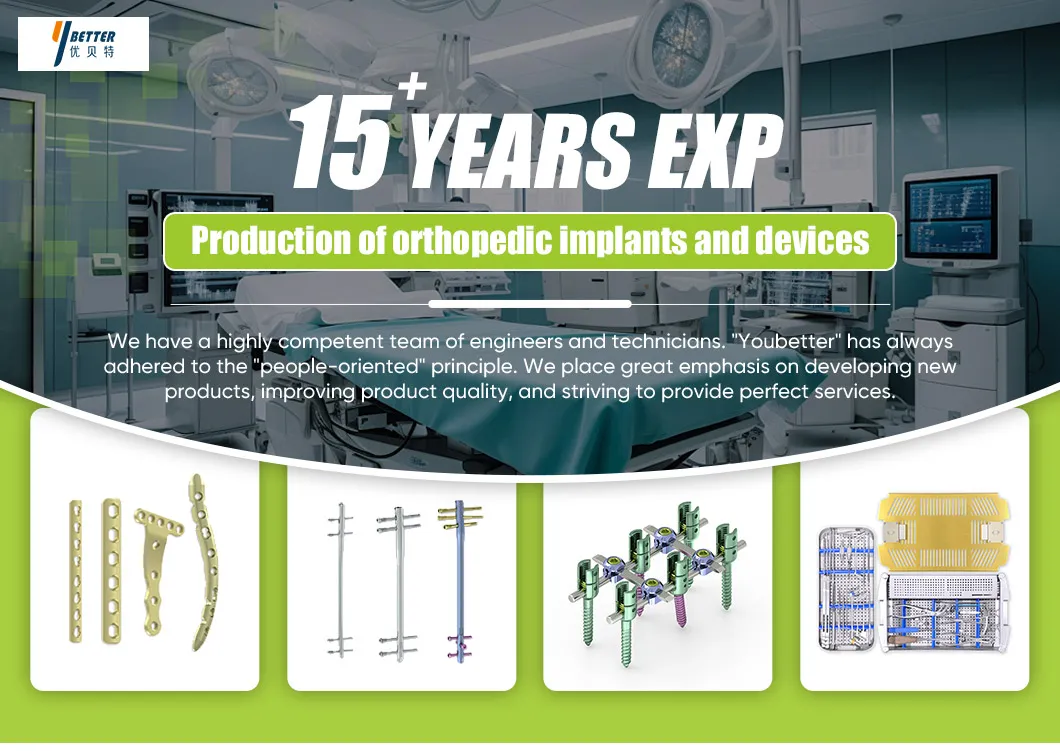
1 / 5










| Still deciding? Get samples of $ ! US$ 50/Piece Request Sample |




| Product Name | Hook Guide Pin |
| Tip Design | Hook-shaped, atraumatic engagement end |
| Material | High-Strength Surgical Stainless Steel (AISI 316L or equivalent) |
| Surface Finish | Polished, smooth, corrosion-resistant |
| Sterilization | Autoclavable (reusable) |
| Diameter Options | Typically 1.5mm, 2.0mm, 2.5mm, 3.0mm |
| Compatibility | Cannulated systems, drilling guides, and fluoroscopy |
| Regulatory | CE Marked, ISO 13485 compliant |

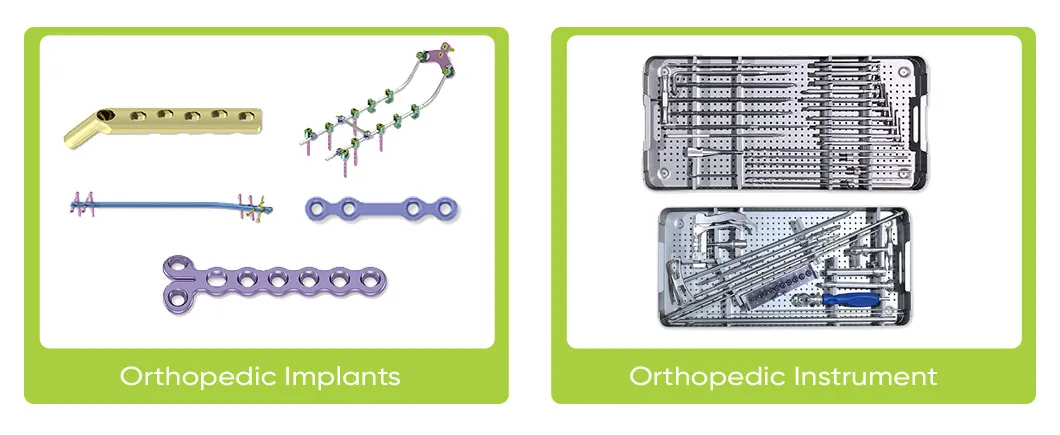


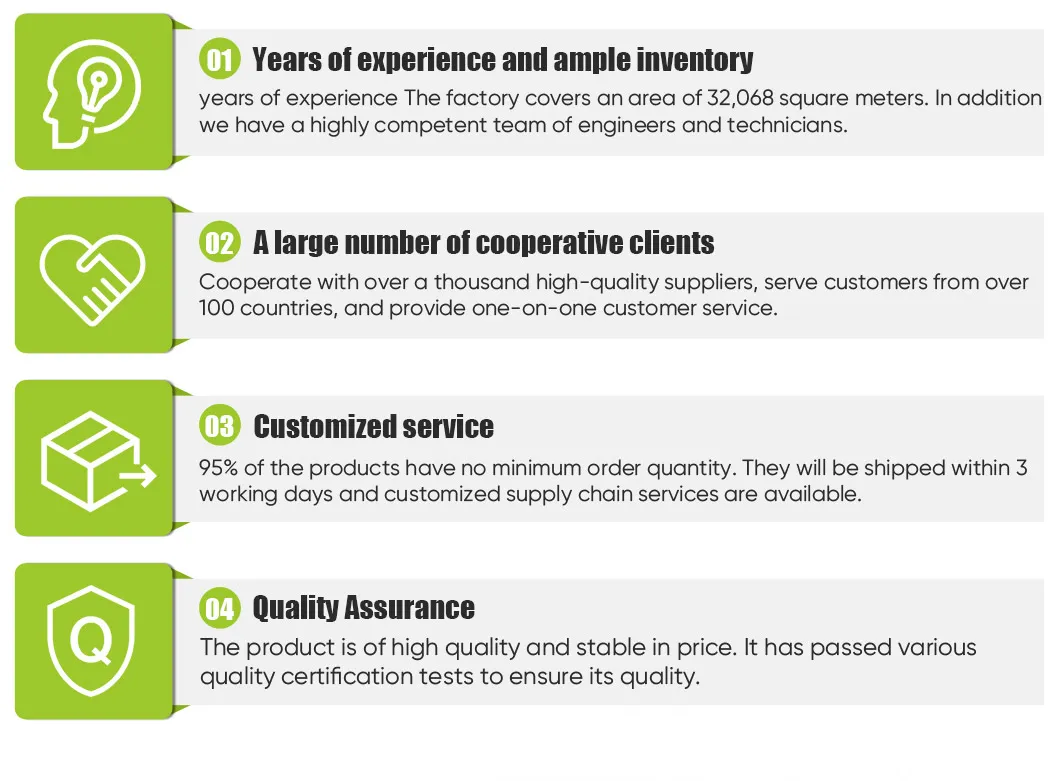


 Weed Orthopedic Implants
Weed Orthopedic Implants