📝 Product Introduction
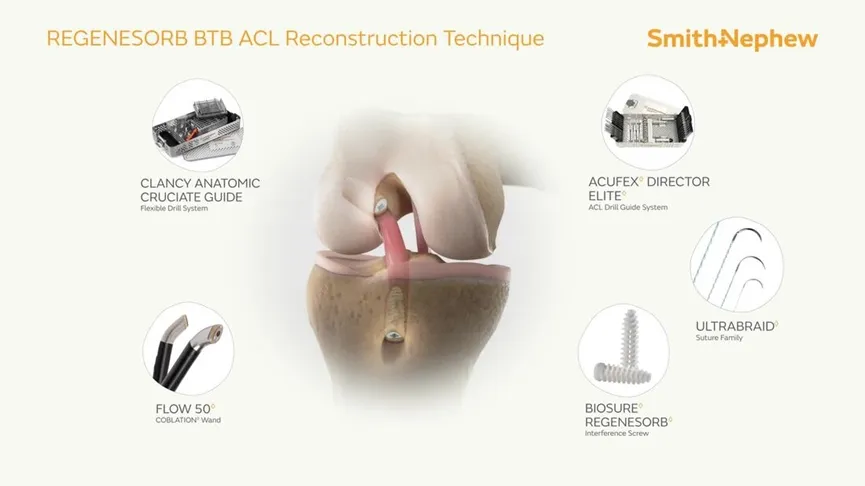
The Interface Screw is designed for use in ACL reconstruction surgeries to secure tendon or ligament grafts in the femoral and tibial bone tunnels. It provides a strong and reliable fixation by compressing the tendon or ligament graft directly against the bone tunnel wall. The screw's unique mechanism ensures optimal stability and a firm attachment between the soft tissue graft and the bone.
The system includes a conical screw, which facilitates easy insertion, and a protective sheath that shields the soft tissue during insertion and reduces the risk of graft displacement or screw breakage. This system is made from PEEK (Polyetheretherketone), a biocompatible material that mimics the elasticity of natural bone.
✅ Advantages & Indications
Indications: This Interface Screw System is primarily used for ACL reconstruction surgeries, providing secure fixation for soft tissue grafts or bone-tendon grafts in the knee joint.
Core Advantages: The PEEK material's natural bone-like properties ensure better integration and flexibility. The long driver slot enhances performance and prevents damage from excessive torque during the fixation process.
❓ Frequently Asked Questions
Q: Why choose this Interface Screw system?
A: Our system is trusted by over 2,000 agents globally. It combines PEEK material biocompatibility with a specialized star drive mechanism for superior fixation and recovery outcomes.
Q: What shipping methods and costs should we expect?
A: We typically utilize courier services for international shipping. Costs are determined by destination, package size, and weight; we provide a detailed quote once requirements are confirmed.
Q: Are the medical products ISO certified?
A: Yes, our production holds the EN ISO 13485 certification, ensuring all devices meet the most stringent safety and performance requirements in the medical industry.
Q: What is the minimum order quantity (MOQ)?
A: Our standard minimum order value is US$1,000. However, sample orders are available at cost for clients needing to evaluate products before a full commitment.
Q: Can you provide products not currently listed in the catalogue?
A: Yes, we accommodate custom orders. By providing pictures or samples, we can work with you to develop similar items, though lead times may vary for custom solutions.
Q: Do you maintain stock for immediate delivery?
A: We maintain stock for most listed products with regular monthly production. Inventory levels vary based on demand, so please consult our team for current availability.












 Weed Orthopedic Implants
Weed Orthopedic Implants