




1 / 5










| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |

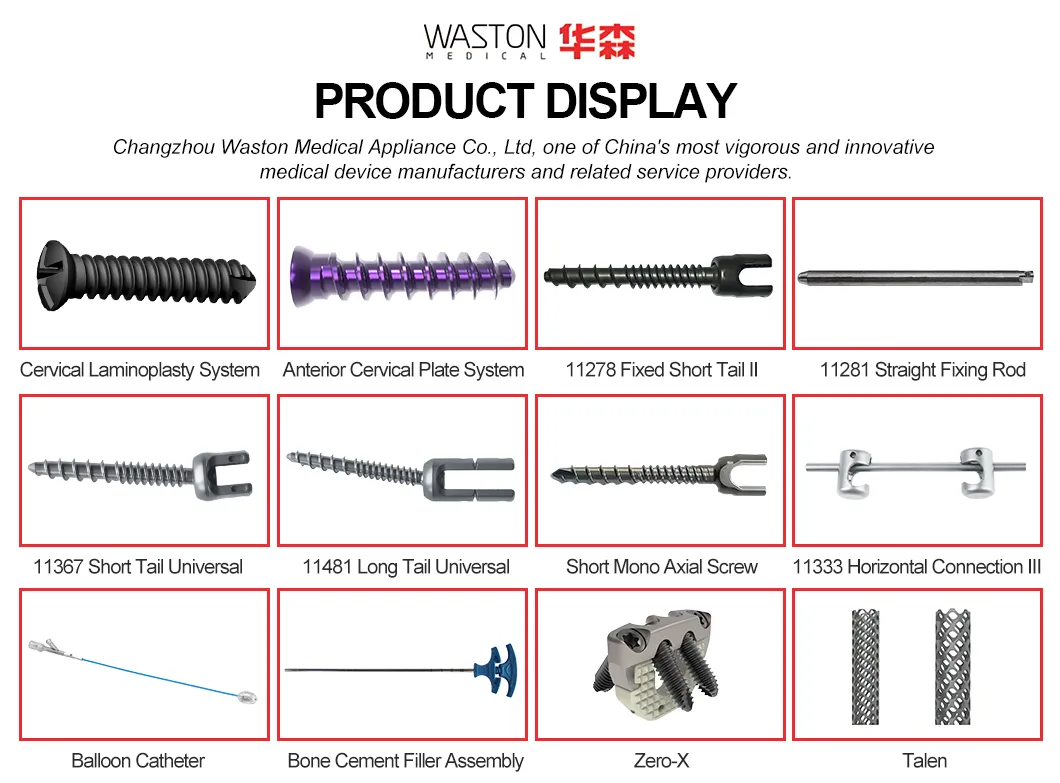
| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | L=8 | ||
| 10511310 | L=10 |


Operations follow strict ISO13485 quality systems and CE MDD 93/42/EEC directives. Production utilizes advanced German, American, and Japanese-imported machining centers and automatic lathes to ensure first-class equipment quality.

The testing center includes biomechanical and physical-chemical laboratories, focusing on raw material procurement, product quality control, and new product performance analysis.
Products are exported to many countries, including France, Turkey, Indonesia, Mexico, Russia, and Thailand. Quality meets international standards as confirmed by global market feedback. We also serve as a major OEM supplier for global industry leaders.



 Weed Orthopedic Implants
Weed Orthopedic Implants