




1 / 5










| Still deciding? Get samples of $ ! US$ 108/Piece Request Sample |
Proximal humeral fractures are among the most common fractures, accounting for 4%–5% of all fractures. Mechanism of injury: Usually occurs when the elbow strikes the ground or the arm is braced during a fall, especially in elderly patients, or when the shoulder joint directly sustains external force.


We are a leading manufacturer of standardized orthopedic implants and instruments for global distributors. Our professional range covers trauma, spine, joints, sports medicine, and CMF — all supported by complete surgical instrument sets.
With in-house R&D, certified laboratories, and modern production lines, we provide reliable, scalable manufacturing focused on high-volume distribution and emerging markets.
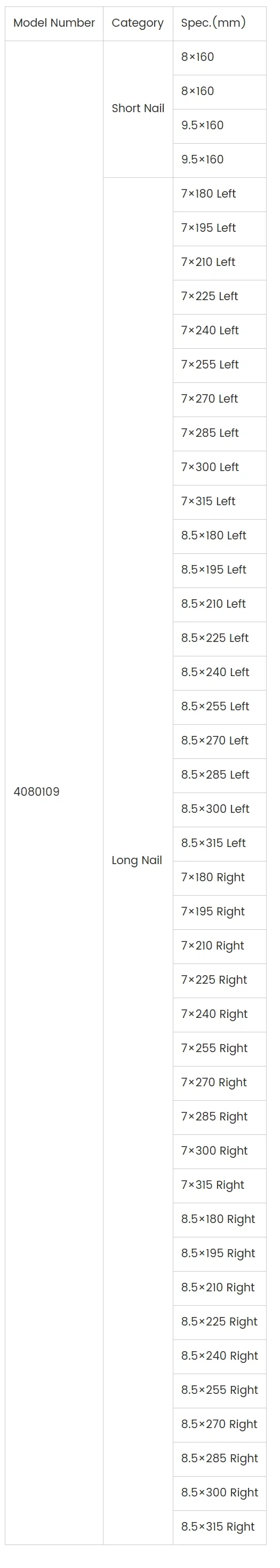
 Weed Orthopedic Implants
Weed Orthopedic Implants