









| Still deciding? Get samples of $ ! US$ 30140/Set Request Sample |
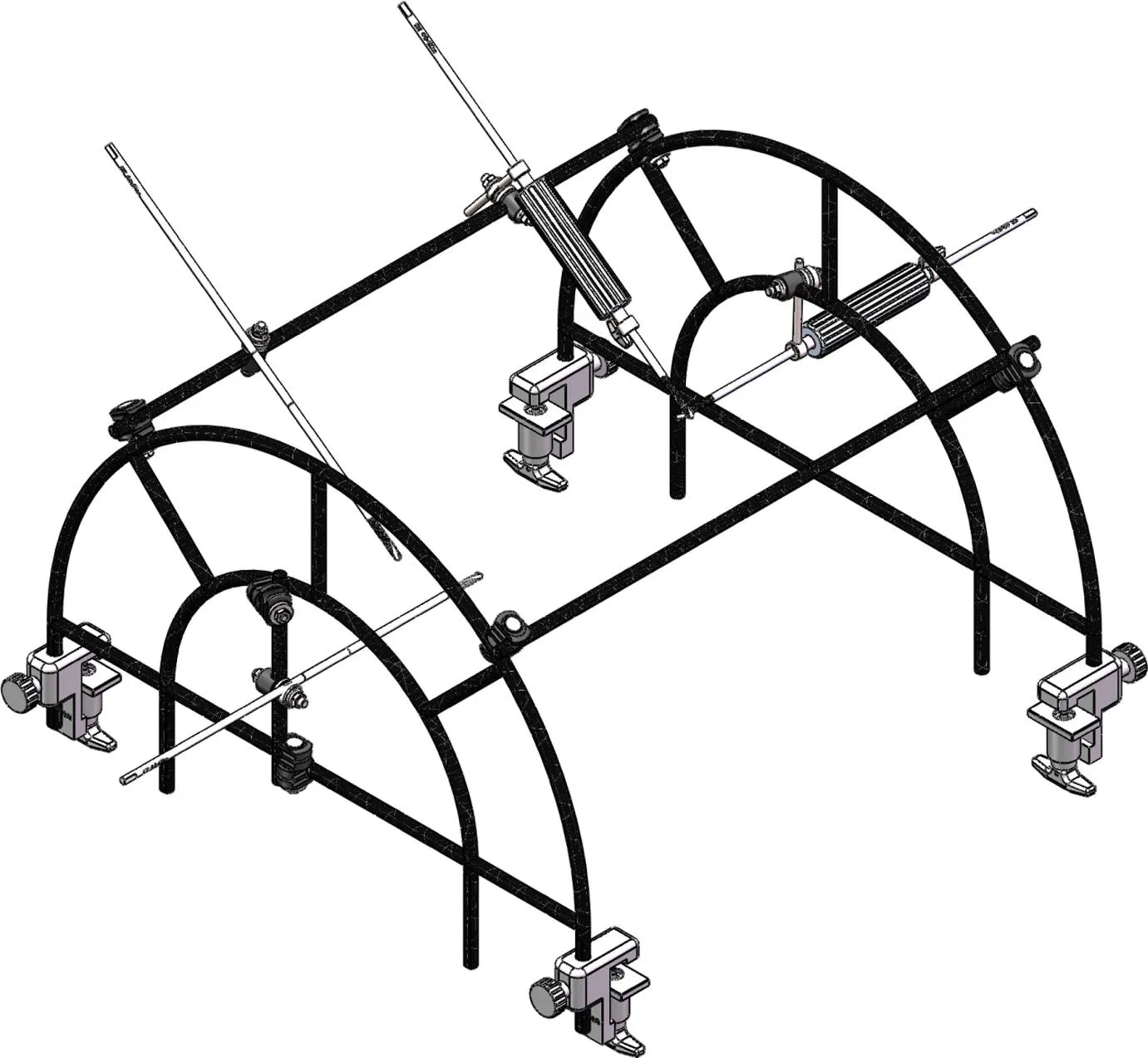
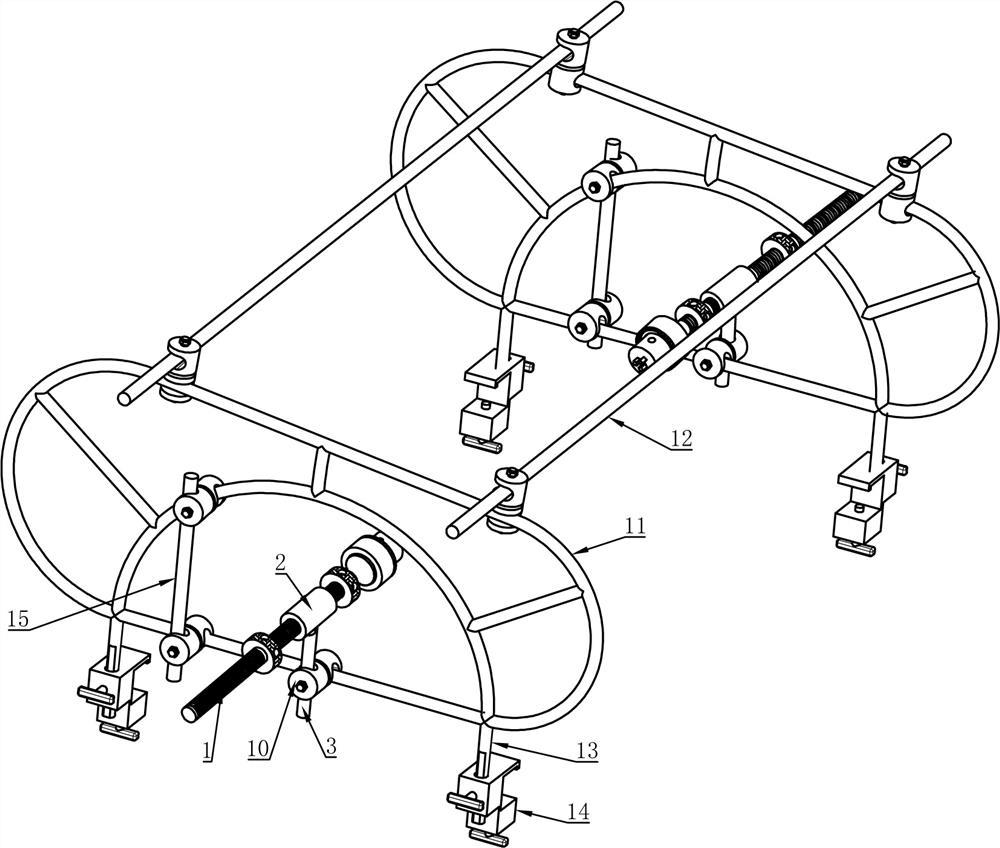
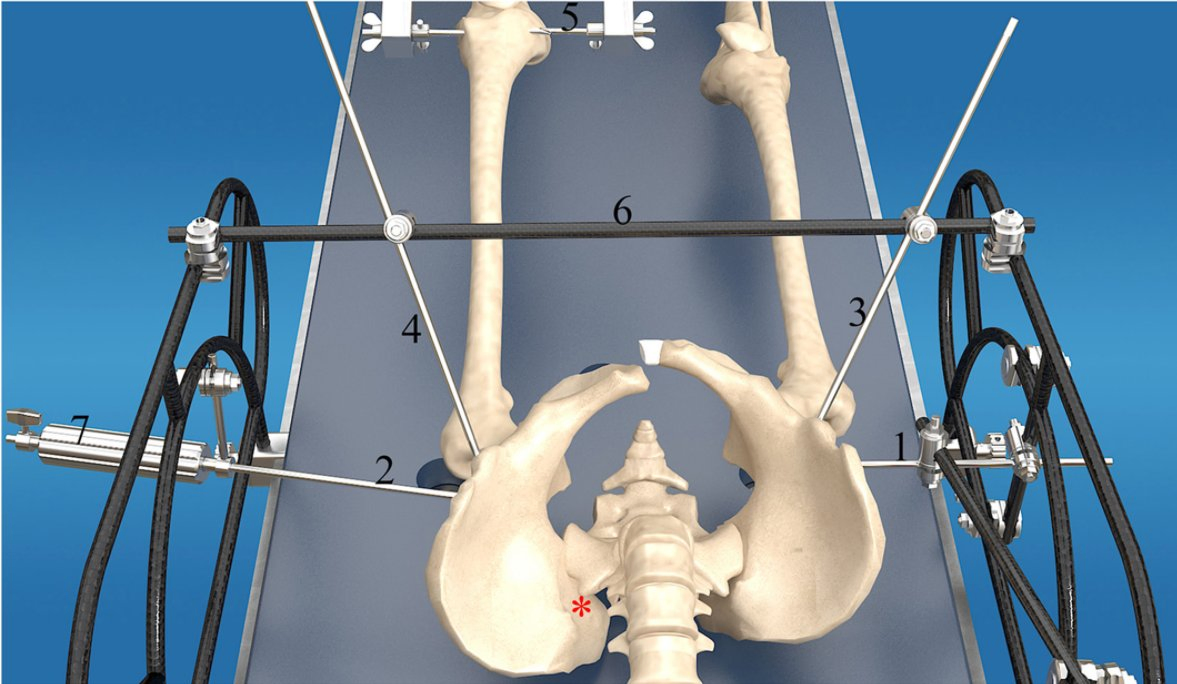
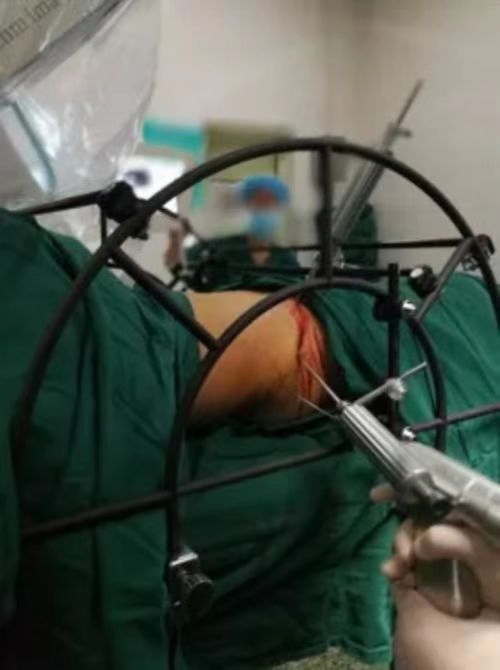
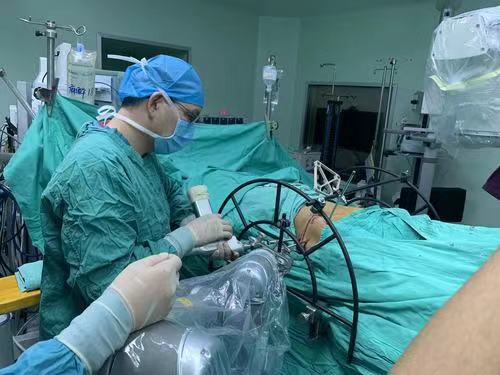
Medical Surgery Orthopedic Pelvic External Fixator Pelvic Reduction Frame
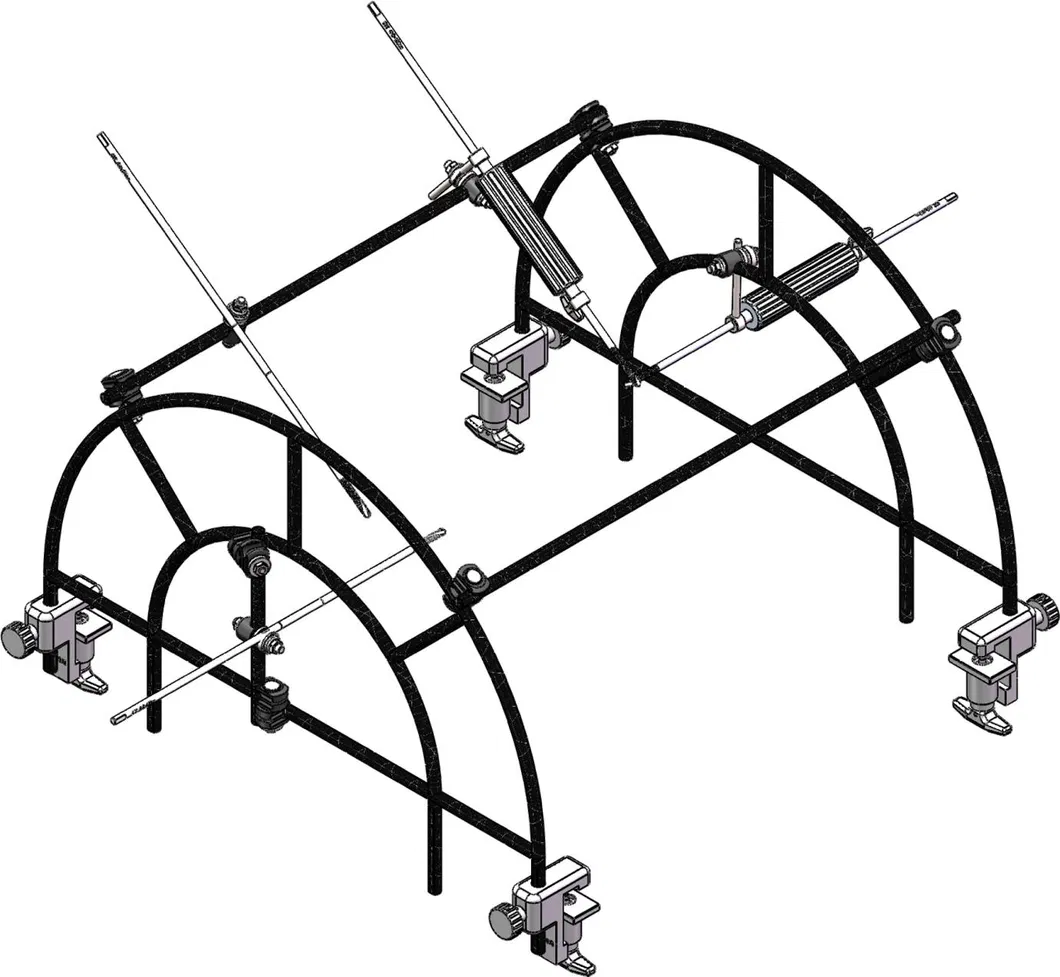
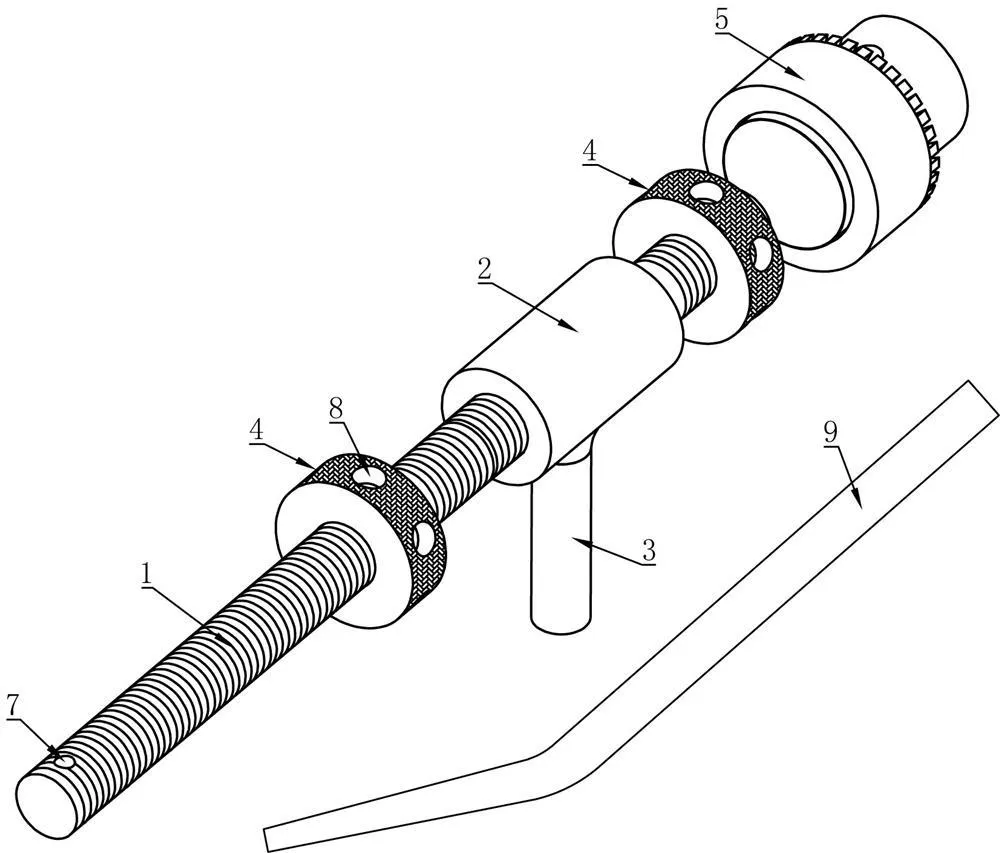
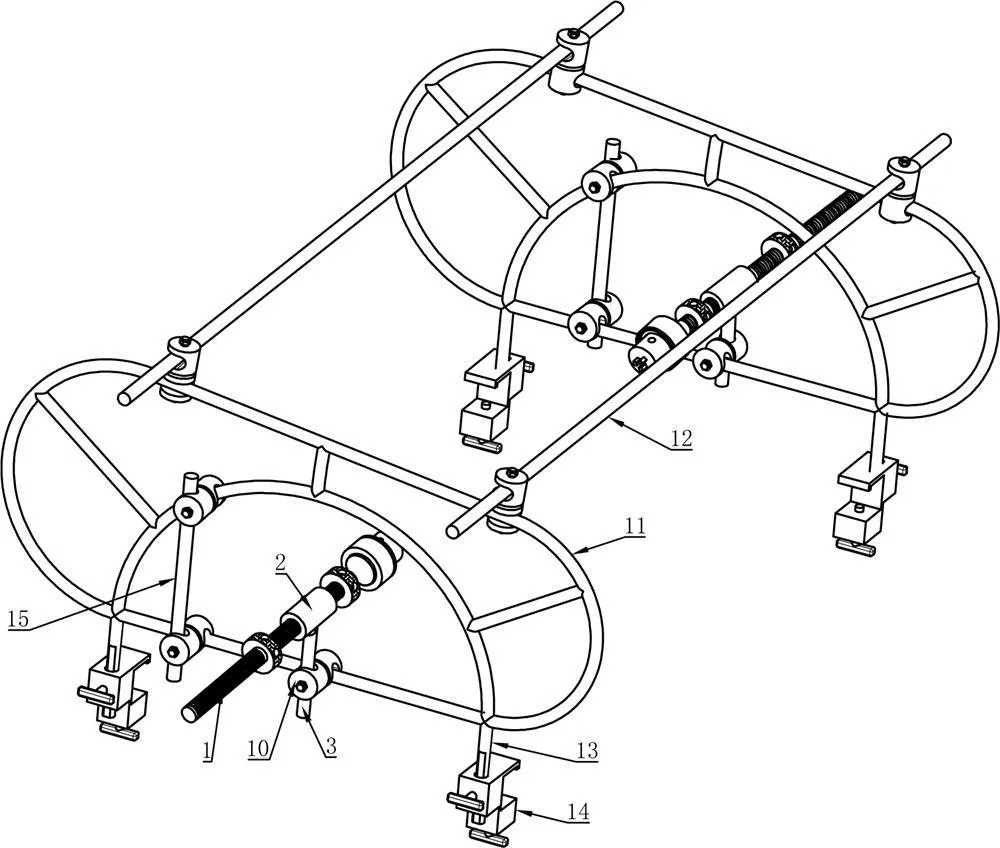
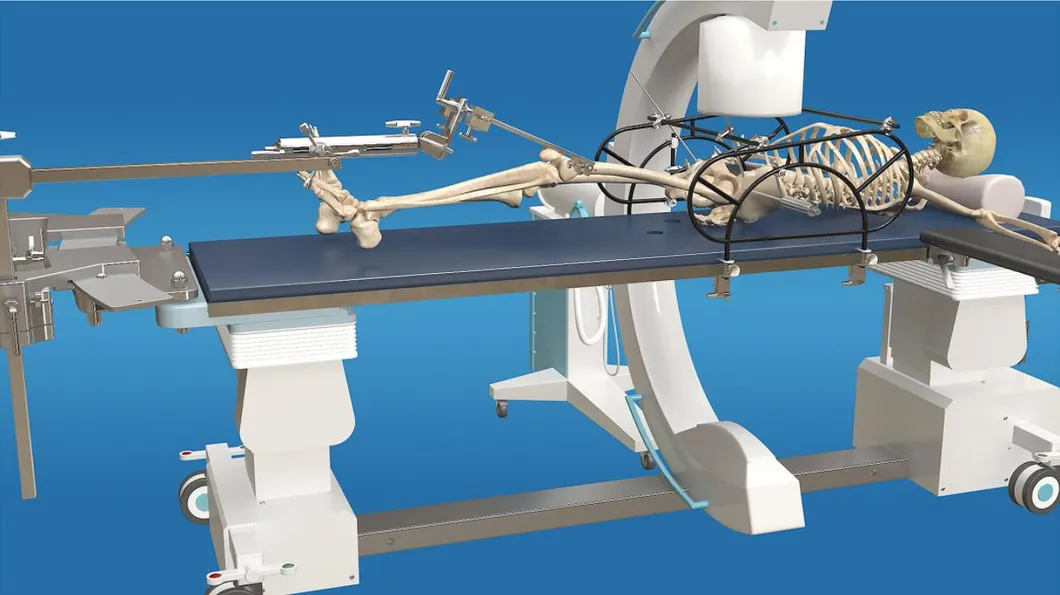
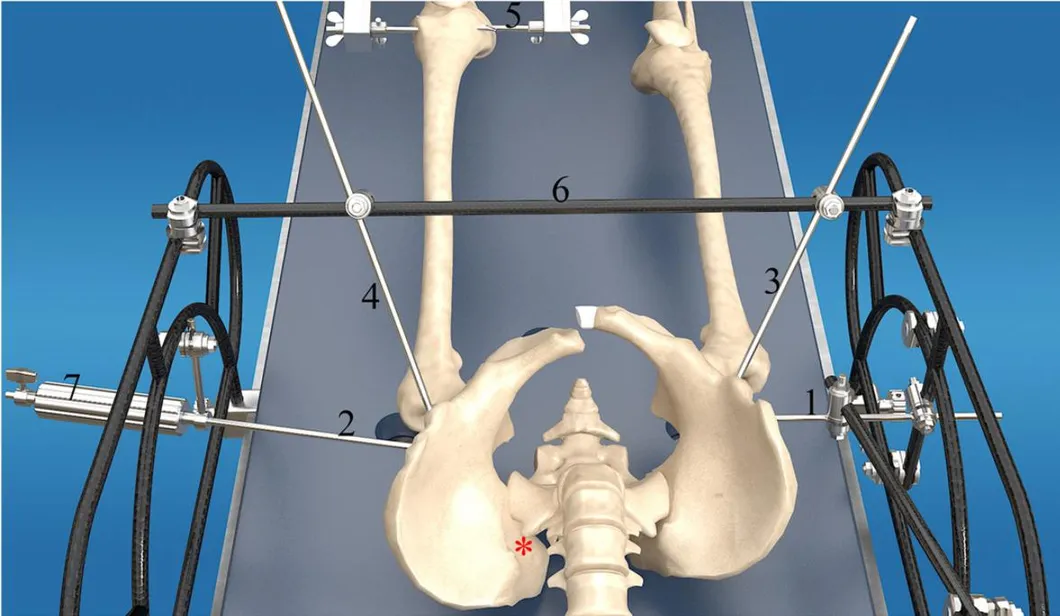
The goal of surgical treatment of pelvic ring disruptions is to restore normal alignment. Direct manipulation of fracture fragments makes reduction easier. However, open reduction and internal fixation of the posterior pelvic ring carry a fairly high wound complication rate. Percutaneous methods of reduction and stabilization carry a far lower wound complication rate, but achieving an adequate re-alignment of the pelvic ring with minimally invasive techniques is difficult.
Manual traction on the injured side often causes the uninjured side to roll and deform, while anterior external fixation usually does little to improve the posterior ring in grossly unstable fractures. Pelvic reduction, whether done via open or closed methods, is an exercise in the application of force to correct fracture deformity. Fracture displacement is almost always multiplanar. Thus, it seems clear that what is needed is a mechanism that allows the surgeon to control and manipulate multiple fracture fragments in multiple planes.
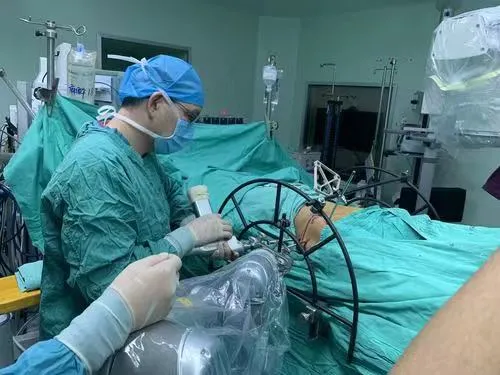
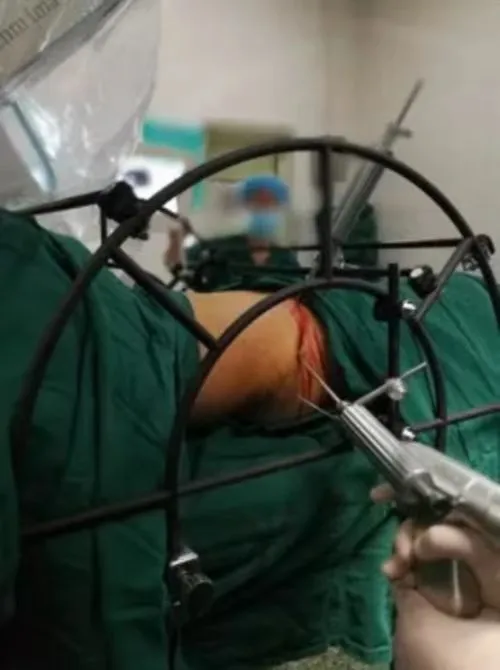

We are a leading manufacturer of high-tech medical equipment used in OR and ICU. Our product range includes Operating Tables, Operating Lights, Anesthesia Machines, Ceiling Pendants, and ICU Ventilators. We advocate resolving technical difficulties comprehensively based on excellent technical R&D to satisfy customer requirements and demand.
Our R&D team has more than 10 years of experience in the line of operating tables. Every year, 10% of turnover is invested in the R&D department. We have achieved a leading position in technical levels through continuous scientific technology innovation, aiming to exceed international standards. We also provide professional OEM and ODM services to global partners through our dedicated mold factory.




 Weed Orthopedic Implants
Weed Orthopedic Implants