Basic Information
Model NO.
External Fixation
Certification
CE, ISO13485
Material Type
Titanium Alloy & Stainless Steel
Production Capacity
500000 Pieces/Year
Product Description
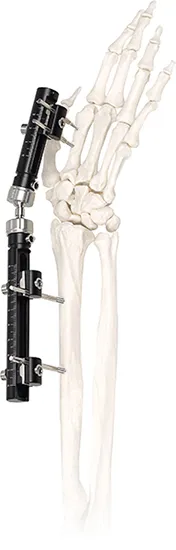
External Fixator-- Unilateral Nondetachable External Fixation System
🛡️
Stable Design: Unilateral fixator, light and stable, specifically applicable for emergency cases.
⏱️
Efficient Operation: Easy to operate, significantly saving surgery time.
🩹
Minimal Invasive: Minimal-invasive surgery with no effect to the blood supply to fracture.
🏥
Easy Removal: No second surgery required; can be safely removed at the clinic.
🦴
Dynamic Design: Consistent with the bone shaft with a controllable dynamic design to promote union.
⚙️
Reliable Screws: Bone screw designed with conical screw thread for a fixed and reliable hold.
Technical Specifications & Variants

| Mini Fixator | 1 Set |
| Bone Screws 2.0*50mm | 4 Pieces |
| Wrench / T-Wrench | 1 Piece Each |

| Mini Fixator (Straight) | 1 Set |
| Bone Screws 2.0*50mm | 4 Pieces |
| Wrench / T-Wrench | 1 Piece Each |

| Adjustable Metacarpal Fixator | 1 Set |
| Bone Screws 2.0*50mm | 4 Pieces |
| Wrench / T-Wrench | 1 Piece Each |

| Lengthening Metacarpal Fixator | 1 Set |
| Bone Screws 2.0*50mm | 4 Pieces |
| Wrench / T-Wrench | 1 Piece Each |

| Cross-articular Metacarpal Fixator | 1 Set |
| Bone Screws 2.0*50mm | 4 Pieces |
| Wrench / T-Wrench | 1 Piece Each |

| Mini Fixator A | 1 Set |
| Bone Screws 2.0*50mm | 4 Pieces |
| Wrench / T-Wrench | 1 Piece Each |

| Mini Fixator B Type | 1 Set |
| Bone Screws 2.0*50mm | 4 Pieces |
| Wrench / T-Wrench | 1 Piece Each |

| Mini Fixator C | 1 Set |
| Bone Screws 2.0*50mm | 4 Pieces |
| Wrench / T-Wrench | 1 Piece Each |
Frequently Asked Questions
What are the primary advantages of this unilateral external fixator?
The system is lightweight, stable, and designed for rapid application in emergency cases. Its minimal-invasive nature preserves the blood supply to the fracture site, promoting faster healing.
Is the external fixation system reusable?
Yes, as specified in the basic information, the feature of this system is that it is reusable after proper sterilization and inspection.
Does the removal of the fixator require another surgery?
No, one of the key benefits is that the fixator can be removed in a clinic setting, eliminating the need for a secondary surgical procedure.
What materials are used for the fixator and screws?
The system is constructed from high-quality Titanium Alloy and Stainless Steel, ensuring biocompatibility and structural integrity.
How does the dynamic design assist in bone union?
The controllable dynamic design allows for micro-movements consistent with the bone shaft, which stimulates callus formation and accelerates the bone healing process.
What certifications do these medical devices hold?
The external fixation systems are certified under CE and ISO13485 standards, meeting international quality and safety requirements for medical devices.























 Weed Orthopedic Implants
Weed Orthopedic Implants