




1 / 5










| Still deciding? Get samples of $ ! US$ 40/Piece Request Sample |
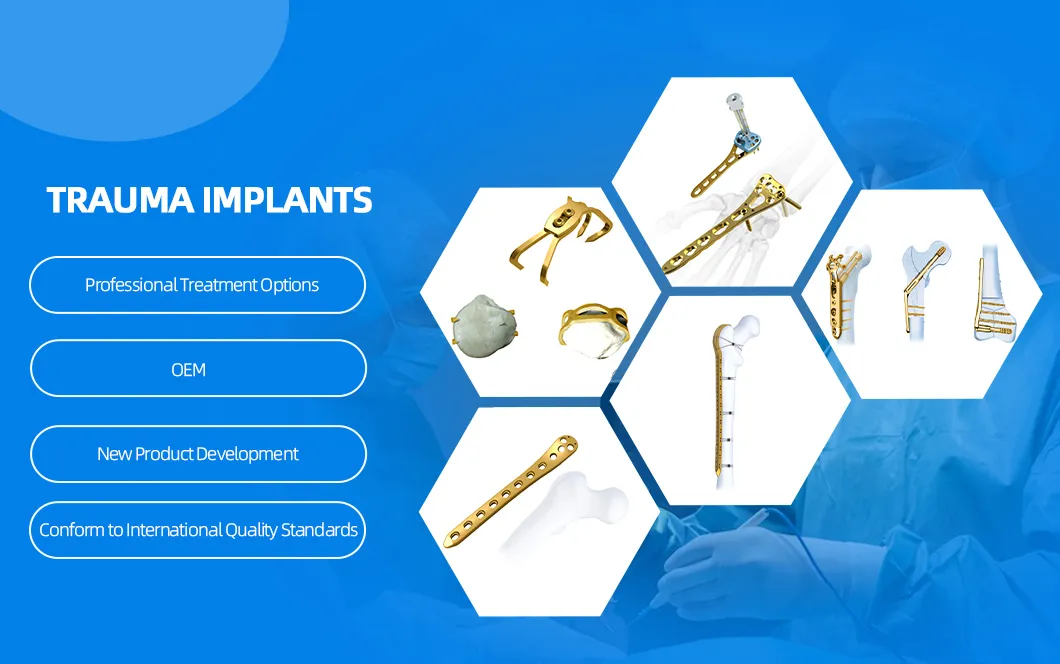


| Product Specification | Length (mm) | Material |
|---|---|---|
| φ3.5mm Cannulated Screw, Hexagonal, Partially Threaded | 14/16/18/20/22/24/26/28/30/32/34/36/38/40/42/44/46/48/50 | SS / Titanium |
| φ4.0mm Cannulated Screw, Hexagonal, Partially Threaded | 14/16/18/20/22/24/26/28/30/32/34/36/38/40/42/44/46/48/50 | SS / Titanium |
| φ4.5mm Cannulated Screw, Hexagonal, Partially Threaded | 30/32/34/36/38/40/42/44/46/48/50/52/54/56/58/60/65/70 | SS / Titanium |






 Weed Orthopedic Implants
Weed Orthopedic Implants