




1 / 5










| Customization: | Available |
|---|---|
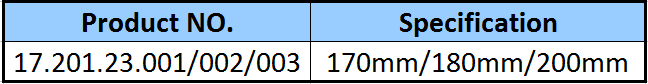
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |





 Weed Orthopedic Implants
Weed Orthopedic Implants