Basic Specifications
Model NO.External Fixation
FeatureReusable
CertificationCE, ISO13485
ColorWhite & Black
UsageBone Fixation
Material TypeTitanium Alloy & Stainless Steel
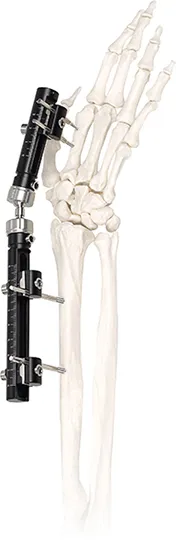
NameExternal Fixator
OriginChina
HS Code90211000
Capacity500,000 Pieces/Year
Specification50*50*50CM
TransportBox
Frequently Asked Questions
What are the main indications for this External Fixation system?
It is primarily used for bone fixation in phalange and metacarpal fractures, especially in emergency cases requiring stable, unilateral support.
What materials are used in the manufacturing of the fixator?
The system utilizes high-grade Titanium Alloy and Stainless Steel to ensure biocompatibility and structural integrity.
Does the patient need a second surgery to remove the fixator?
No, the design allows for easy removal at a clinic, eliminating the need for a second surgical procedure and reducing patient recovery stress.
How does the "dynamic design" benefit the healing process?
The controllable dynamic design allows for micro-movements consistent with the bone shaft, which is clinically proven to promote bone union and faster healing.
Is the external fixator reusable?
Yes, the device is labeled as reusable, provided it undergoes proper sterilization and inspection protocols between uses.
What quality certifications does this product hold?
The system is fully certified with CE and ISO13485 standards, meeting international requirements for medical devices.






















 Weed Orthopedic Implants
Weed Orthopedic Implants