













| Still deciding? Get samples of $ ! US$ 2.5/Piece Request Sample |
| Model NO. | WZ-TB-203 |
| Sterilization | Ethylene Oxide Sterilization |
| Quality Guarantee Period | Two Years |
| Application | Orthopedic Surgery |
| Usage | Single-Use |
| Specification | D: 11mm, 14mm, 20mm |
| Origin | China |
High Quality CE ISO Certified Cruciate Ligament Circle Titanium Button - Factory Manufactured Endobutton. This product is designed specifically for orthopedic surgery, ensuring high-strength suspension fixation.

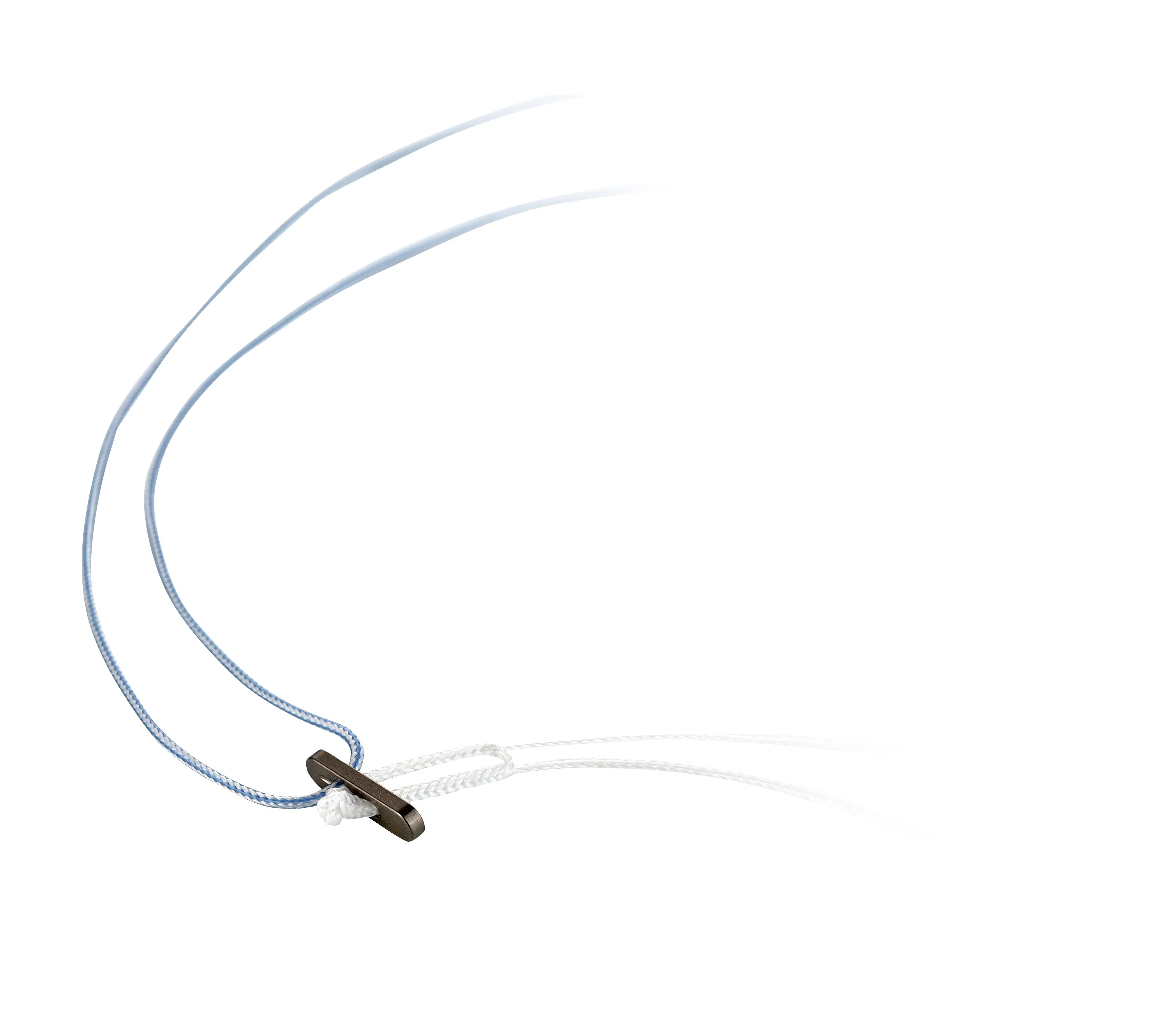
Circle Titanium Button I-WZ-TB-203
Use with WZ-TB-301 Loop. Various sizes available: D:11.0mm, 14.0mm, 20.0mm.
| Name | Description | Model | Size |
|---|---|---|---|
| Titanium Button with Loop I | Suitable for cruciate ligament reconstruction. Loop can be adjusted freely, improve tendon bone healing. | WZ-TB-101 | D: 3.5mm |
| Titanium Button II | Suitable for cruciate ligament reconstruction. Extensive converage of cortical bone. High-strength suspension fixation. | WZ-TB-201 | D: 8.0mm |
| Revision Titanium Button I | Use with WZ-TB-101 Adjustable Titanium Button. Better suspension fixation. | WZ-TB-202 | D: 3.5mm |
| Circular Titanium Button I | Use with WZ-TB-301 Loop. | WZ-TB-203 | D: 11.0mm, 14.0mm, 20.0mm |
| Loop | f(min): 15.0mm | WZ-TB-301 | f(min): 15.0mm |


To better ensure the safety of your goods, professional, environmentally friendly, convenient and efficient packaging services will be provided in sterilization blister bags.

Established in Sept. 2004, our manufacturing facility is located in Zhejiang Province. We are mainly engaged in the development, manufacture, and sales of CE certified high-end medical supplies used in the operating room and emergency room. We also undertake OEM orders of medical products. Our sales network covers the European Union, the Middle East, Southeast Asia, South Korea, and Africa. We are committed to high quality products and excellent services to meet the demands of global customers.



Certification: ISO 13485 & CE MDD Approved facility.

We perform a pre-production sample check before mass production and a final comprehensive inspection before every shipment to ensure consistency and quality.
Our products are ISO 13485 and CE MDD approved, meeting international standards for orthopedic surgical instruments.
Yes, we undertake OEM orders for medical products to meet the specific requirements of our global clients.
It is primarily used for cruciate ligament reconstruction in orthopedic surgeries, providing high-strength suspension fixation for bone-tendon healing.
The buttons are made of high-grade Titanium and are sterilized using Ethylene Oxide (EO) to ensure they are ready for single-use surgical applications.
The products have a quality guarantee period of two years. They should be stored in their original sterilization blister bags until use.
 Weed Orthopedic Implants
Weed Orthopedic Implants