


1 / 5










| Still deciding? Get samples of $ ! US$ 149/Set Request Sample |
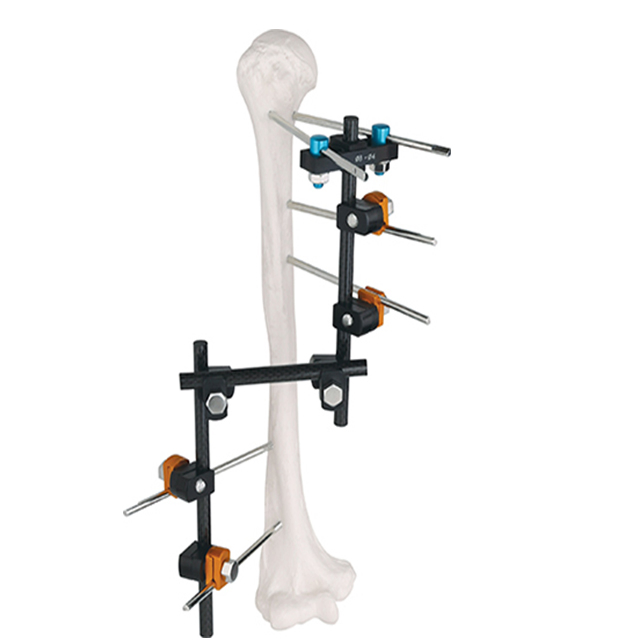
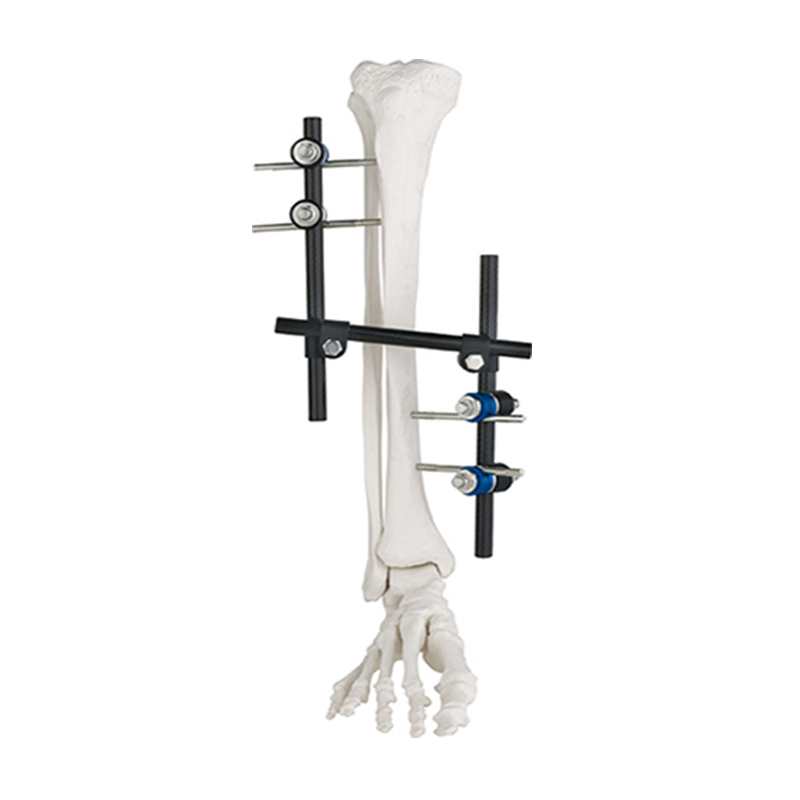
| Product Number | Product Name | Assembly Part | Part ID | Size | Qty |
|---|---|---|---|---|---|
| ZAS001 | T frame | Pin to rod coupling | ZASP01 | φ11/5-6 | 4 |
| T clamp | ZJYP02 | φ11/5 | 1 | ||
| Connecting rod | ZASL01 | φ11×300 | 1 | ||
| Standard screw | SS10501345 | HA5×130 | 4 | ||
| Standard screw | SS10501550-B | HB5×150 | 2 | ||
| Used for: Femur, Tibia | |||||






 Weed Orthopedic Implants
Weed Orthopedic Implants