Turkey has rapidly emerged as a global hub for medical excellence, particularly in the field of orthopedics. As a bridge between Europe and Asia, the Revision Hip/Knee Systems Manufacturers in Turkey are witnessing an unprecedented surge in demand. This growth is driven by a combination of high-caliber surgical expertise, a robust medical tourism sector centered in cities like Istanbul, Ankara, and Izmir, and a manufacturing infrastructure that adheres to stringent EU standards.
The current industrial status in Turkey reflects a transition from importing high-end medical devices to local high-precision manufacturing. Turkish manufacturers and international collaborators are focusing on revision arthroplasty, which is significantly more complex than primary joint replacement. With an aging global population and the increasing longevity of patients who received primary implants decades ago, the need for sophisticated revision systems—capable of addressing bone loss, infection, and implant instability—has never been higher.

The integration of Artificial Intelligence (AI) and 3D printing is revolutionizing how revision surgeries are performed in Turkey. Local manufacturers are increasingly utilizing 3D-printed titanium implants with trabecular structures that mimic natural bone. These porous surfaces enhance biological fixation, which is crucial in revision cases where bone quality is compromised.
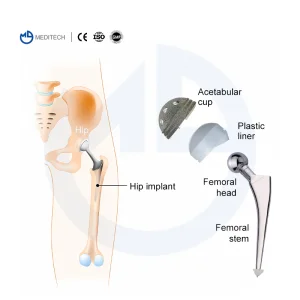
From Shape Memory Alloys to high-crosslinked polyethylene (XLPE), Turkish medical device manufacturers are prioritizing materials that reduce wear and tear. Revision Hip Systems now often feature modular necks and stems to allow surgeons to customize the fit to the patient's specific anatomy, addressing the unique challenges of bone defects found in Turkey's aging patient demographic.
Robot-assisted revision surgery is gaining traction in Istanbul's private hospitals. This trend necessitates Revision Knee Systems that are compatible with navigation software, ensuring sub-millimeter precision. Turkish manufacturers are responding by developing instrument sets that are optimized for digital surgical planning.
The business model for Revision Hip/Knee Systems Manufacturers in Turkey is highly collaborative. Companies like Shenzhen Weed Medical Apparatus Co., Ltd. provide a blueprint for this success: bridging the gap between high-tech manufacturing and frontline clinical needs.
In the Turkish market, success is determined by two main factors: Direct Hospital Engagement and a Robust Distribution Network. Turkish healthcare providers, including both the Social Security Institution (SGK) and private groups like Acıbadem or Memorial, demand products that are CE certified and proven to reduce the "length of stay" for patients.
Our collaboration with professors and clinical staff ensures that our revision systems are not just products, but clinical solutions. In Turkey, the feedback loop from the surgery room to the factory floor is short, allowing for rapid iterations of instruments like the PFNA Instrument Set or S-ROM Titanium Alloy Hip Joints.
Shenzhen Weed Medical Apparatus Co., Ltd., established in 2015, primarily engages in the sales of medical devices and maintains in-depth business and technical cooperation with leading domestic and international consumable manufacturers. Most of its products boast technology and materials superior to industry standards.
The company's business is divided into direct sales and distribution. Its products include absorbable materials, shape memory alloys, 3D-printed orthopedic consumables, external fixators, and other major surgical consumables. These products are used by over 200 hospitals in Sichuan Province, and are now being positioned for the high-growth markets of Turkey and the Middle East. The company enjoys a good reputation among peers and medical institutions.
The company collaborates closely with professors, universities, and frontline clinical staff in domestic hospitals. Sales are the company's cash cow. During the sales process, the company consistently focuses on the technological contributions and shortcomings of its products to the industry. The company's technical and sales personnel always pay attention to the user needs, opinions, and suggestions during clinical use. The company's technical department collaborates with hospital experts, university professors, frontline users, and manufacturers to develop solutions to common problems.
As Revision Hip/Knee Systems Manufacturers in Turkey continue to innovate, the focus is shifting toward "Smart Implants." These future systems will likely include sensors to monitor intra-articular pressure and early signs of infection. For Turkish clinics specializing in complex revisions, these advancements mean higher patient satisfaction and lower re-operation rates.
Furthermore, the Turkish Medical Device Regulatory Framework (aligned with MDR) ensures that every product, from the Proximal Lateral Femoral Plate to the Artificial Knee Joint Implants, meets the highest safety benchmarks. This makes Turkey not only a consumer of these high-tech systems but a leading exporter to Europe and the MENA region.
Our commitment remains steadfast: to supply the Turkish market with medical devices that incorporate AI-driven design and next-generation materials. By choosing our revision systems, Turkish hospitals are investing in durability, precision, and patient-centric care.