 Total Hip Replacement Prosthesis Corail Femoral Stem
Total Hip Replacement Prosthesis Corail Femoral Stem
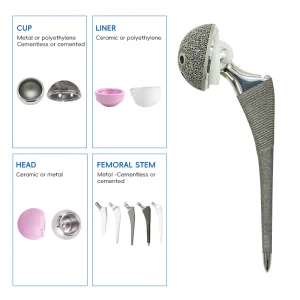 UHMWPE Insert Acetabular Cup System
UHMWPE Insert Acetabular Cup System
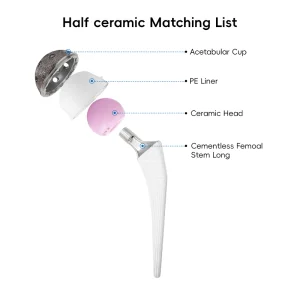 Orthopedic Prosthesis Artificial Hip and Knee
Orthopedic Prosthesis Artificial Hip and Knee
 Total Knee Joint Replacement Instrument Set
Total Knee Joint Replacement Instrument Set
 China Factory Orthopedic Knee Implants
China Factory Orthopedic Knee Implants
 Instruments for Knee Replacement Surgery
Instruments for Knee Replacement Surgery
 Hip Joint Prosthesis Acetabular Cup
Hip Joint Prosthesis Acetabular Cup
 Hip Joint CE Prosthesis Replacement
Hip Joint CE Prosthesis Replacement
The global elbow joint prosthesis market is entering a phase of rapid evolution. Historically overshadowed by hip and knee replacements, total elbow arthroplasty (TEA) is now gaining significant traction. Current industrial standards demand high-precision biomechanical replication, shifting from simple "hinge" designs to advanced semi-constrained and unconstrained systems that allow for natural rotational freedom.
Manufacturers are navigating a complex landscape of CE MDR (Medical Device Regulation) and FDA Class III requirements. The focus has shifted towards clinical longevity and wear-rate reduction. As a leading manufacturer, we align our production lines with these rigorous global safety benchmarks to ensure patient safety across continents.
The industrial status of elbow prosthetics is characterized by a high barrier to entry. The complex anatomy of the elbow—involving the humerus, ulna, and radius—requires implants that can withstand multi-axial stress while maintaining stability. Current commercial data indicates a steady 5.8% CAGR in the upper extremity implant market, driven by post-traumatic reconstruction and an aging population suffering from rheumatoid arthritis.
The industry is moving away from "one-size-fits-all" solutions. 3D Printing (Additive Manufacturing) is the core trend, allowing for trabecular bone-like structures that promote faster osseointegration. AI-driven surgical planning software now enables surgeons to simulate the range of motion before the first incision is made.
Material science is also evolving. We are seeing a transition from traditional Cobalt-Chrome alloys to Titanium-Niobium coatings and advanced UHMWPE (Ultra-High Molecular Weight Polyethylene) liners that virtually eliminate wear debris, significantly extending the lifespan of the prosthesis.
International buyers, from government healthcare tenders to private surgical clinics, are looking for more than just a product; they are looking for supply chain resilience and technical support. Key procurement priorities include:
Hospitals require reliable OEM partners who can deliver large batches of standardized implants while maintaining the flexibility for ODM (Original Design Manufacturing) projects tailored to specific ethnic anatomical variations.
Traceability is non-negotiable. Every prosthesis must be accompanied by comprehensive documentation, including material certificates, sterilization validation, and clinical trial summaries.
Modern procurement often bundles implants with specialized instrument sets. Our "implant + tool" integrated solution reduces surgical error and increases operating room efficiency.
As a leading Chinese factory, we combine cost-efficiency with world-class R&D. The "Made in China" label in the medical field now represents high-tech precision. Our advantages include:
Shenzhen Weed Medical Apparatus Co., Ltd., established in 2015, primarily engages in the sales of medical devices and maintains in-depth business and technical cooperation with leading domestic and international consumable manufacturers. Most of its products boast technology and materials superior to industry standards.
The company's business is divided into direct sales and distribution. Its products include absorbable materials, shape memory alloys, 3D-printed orthopedic consumables, external fixators, and other major surgical consumables. These products are used by over 200 hospitals in Sichuan Province. The company enjoys a good reputation among peers and medical institutions.

The company collaborates closely with professors, universities, and frontline clinical staff in domestic hospitals. Sales are the company's cash cow. During the sales process, the company consistently focuses on the technological contributions and shortcomings of its products to the industry. The company's technical and sales personnel always pay attention to the user needs, opinions, and suggestions during clinical use. The company's technical department collaborates with hospital experts, university professors, frontline users, and manufacturers to develop solutions to common problems.
Elbow joint prostheses are utilized across diverse clinical environments. Our products are engineered to excel in:
In high-velocity accident cases where the distal humerus is non-reconstructible, our rapid-response OEM kits provide surgeons with the immediate components needed for complex reconstruction.
For elderly patients with severe osteoporosis and comminuted fractures, our cement-fixed elbow systems offer immediate stability and early mobilization capabilities.
Elite athletic clinics utilize our low-friction, unconstrained designs for young, active patients who require high-performance joint articulation post-injury.